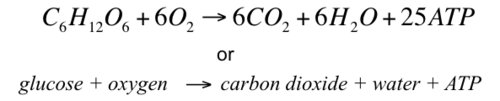Cellular Respiration
Cellular respiration is the set of the metabolic reactions and processes that take place in the cells of organisms to convert biochemical energy from nutrients into adenosine triphosphate (ATP), and then release waste products. The reactions involved in respiration are catabolic reactions, which break large molecules into smaller ones, releasing energy in the process as weak so-called "high-energy" bonds are replaced by stronger bonds in the products. Respiration is one of the key ways a cell gains useful energy to fuel cellular activity. The overall reaction is broken into many smaller ones when it occurs in the body, most of which are redox reactions themselves.
Nutrients that are commonly used by animal and plant cells in respiration include sugar, amino acids and fatty acids, and a common oxidizing agent (electron acceptor) is molecular oxygen (O2). The energy stored in ATP (its third phosphate group is weakly bonded to the rest of the molecule and is cheaply broken allowing stronger bonds to form, thereby transferring energy for use by the cell) can then be used to drive processes requiring energy, including biosynthesis, locomotion or transportation of molecules across cell membranes.
Cellular respiration and fermentation produce energy for cells to use. Any chemical process that yields energy is known as a catabolic pathway. For nearly all organisms on Earth (except chemolithotrophs), that energy is stored in organic molecules. Cells release the energy in those organic molecules by breaking them down. Through cellular respiration and fermentation, those bonds are broken releasing the potential energy of organic molecules into kinetic energy that cells use to do work.
ATP
Adenosine Triphosphate is commonly known as ATP. It is a lot like a chemical spring that gets loaded, and move around a cell, and can be split apart producing energy. ATP is then split into ADP (which is now relaxed) and a phosphate molecule.
ATP consists of adenosine and three phosphate groups (triphosphate). ATP is an unstable molecule in water, in which it hydrolyses to ADP and phosphate. This is because the strength of the bonds between the phosphate groups in ATP are less than the strength of the hydrogen bonds, between its products (ADP + phosphate), and water. Thus, if ATP and ADP are in chemical equilibrium in water, almost all of the ATP will eventually be converted to ADP. A system that is far from equilibrium is capable of doing work. Living cells maintain the ratio of ATP to ADP at a point ten orders of magnitude from equilibrium, with ATP concentrations a thousandfold higher than the concentration of ADP. This displacement from equilibrium means that the hydrolysis of ATP in the cell releases a large amount of free energy.
General Equation for Cellular Respiration
In general, aerobic respiration is the breakdown of food in the presence of oxygen producing carbon dioxide, water and synthesizing ATP. Food can be sugars, starches, or fats. All eukaryotic organisms can convert use glucose as food.
This is a balanced equation of the cellular respiration of glucose. A glucose molecule combines with 6 oxygen molecules, producing 6 molecules of water, 6 molecules of water and ATP.
Cellular Respiration as a Series of Reduction-Oxidation Reactions
Redox describes all chemical reactions in which atoms have their oxidation state changed. The term comes from the two concepts of reduction and oxidation. Whereas, oxidation is the loss of electrons by a molecule or atom. An oxidation of an atom creates a cation, a positively charged ion. Just think about it being a double negative (like in algebra). A negative times a negative creates a positive charge.
In contrast, Reduction is the gain of electrons by a molecule or atom. By gaining electrons, it creates a positive charge on that atom or electron. A simple redox reaction is the ionic bonding of salt (NaCl), where a sodium loses an electron to chlorine generating a positive charged sodium and a negatively charged chlorine. And this is how living things generate usable energy from energy stored in molecules. For every reduction, there is an oxidation. Hence, these reactions are known as redox reactions.
Redox of Glucose
Glucose goes through the same type of reaction. Each carbon atom loses electrons when it is oxidized in the presence of oxygen. And oxygen becomes reduced, because it gains electrons. In this way energy is released from the glucose in small amounts. Living organism can now use this energy to do work. In cells, glucose is oxidized through a long series of carefully controlled redox reactions. The resulting change in free energy is used to synthesize ATP from ADP and P. Together, these reactions comprise cellular respiration.
Energy in cells is generated by moving electrons from one chemical to another. In this way energy in food molecules (like glucose) are released gradually instead of big bursts. A lot like methane in combustion of natural gas, NAD+ is the molecule of cells are the oxidizing agents. As glucose is decomposed it strips electrons from it in order to combine NAD+ with a hydrogen, synthesizing NADH. NADH is like a loaded spring. It has stored energy, trapped in small energy doses in the form of single hydrogen bonds.
Steps of Cellular Respiration
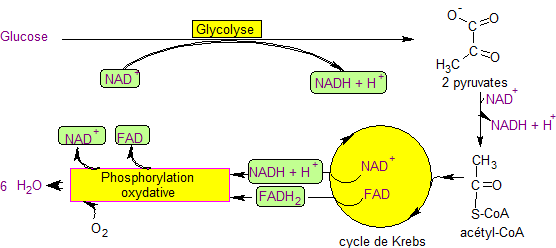
There are three steps of cellular respiration and they always occur in this order: Glycolysis, the Kreb’s cycles, and the Electron Transport Chain, all which go through a series of redox reactions.
Glycolysis
Glycolysis is a metabolic pathway that occurs in the cytosol of the cell and splits glucose into two other molecules, called pyruvate. The free energy released in this process is used to form the high-energy compounds ATP AND NADH. Glycolysis occurs in nearly all organisms, both aerobic (those that utilize oxygen) and anaerobic (those that don’t utilize oxygen). The wide occurrence of glycolysis indicates that it is one of the most ancient known metabolic pathways. Splitting glucose costs the cell 2 ATP, but it gains 4 ATP and releases 2 NADH. As the saying goes, you gotta have money to make money. You gotta have energy to make energy.
Kreb's Cycle (The Citric Acid Cycle)
All aerobic organisms take this process 2 steps further. They take those two pyruvate molecules and break them down a bit further in the Kreb’s cycle. In eukaryotes, the Kreb’s cycle occurs in the matrix of the mitochondrion. The details are quite intricate, but we are going to keep to generalizations. Those 2 pyruvated molecules that were split from glucose in glycolysis move to the mitochondria in eukaryotes (they stay in the cytosol of aerobic prokaryotes), and get further broken down into several pint size energy packets. It produces 2 ATP directly. But it also oxidizes NAD+ (and a similar molecule FAD+) to form NADH and FADH2. The latter molecules are the star of the next stage, the electron transport chain.
Electron Transport Chain
An electron transport chain (ETC) couples electron transfer between an electron donor(such as NADH) and an electron acceptor (such as O2) with the transfer of H+ ions(protons) across a membrane. The resulting electrochemical proton gradient is used to generate chemical energy in the form of (ATP). At the mitochondrial inner membrane, electrons from NADH and FADH2 pass through the electron transport chain to oxygen, which is reduced to water. In other words, the energy locked up in the NADH and FADH2 molecules from glycolysis and the Kreb’s cycle gets released in order to phosphorylate ATP from ADP and a phosphorous molecule.
The electron transport chain comprises an enzymatic series of electron donors and acceptors. Each electron donor passes electrons to a more electronegative acceptor, which in turn donates these electrons to another acceptor, a process that continues down the series until electrons are passed to oxygen, the most electronegative and terminal electron acceptor in the chain. Passage of electrons between donor and acceptor releases energy, which is used to generate a proton gradient across the mitochondrial membrane by actively “pumping” protons into the intermembrane space, producing a thermodynamic state that has the potential to do work. The entire process is called oxidative phosphorylation, since ADP is phosphorylated to ATP using the energy of hydrogen oxidation in many steps. The end product of this is a net of 32 ATP. This is how almost all of the energy of life comes to be.
FERMENTATION
Like respiration, fermentation is the process of extracting energy from the oxidation of organic compounds (like glucose). However, fermentation can occur in the presence or absence of oxygen. Fermentation involves the step of glycolysis in respiration, but it does not go through the Kreb’s cycle or the Electron Transport Chain. Therefore there are only 2 net ATP generated, not the net of 32 that come from cellular respiration. So fermentation produces energy, however, it is 16 times less efficient than cellular respiration.
Overview of ethanol fermentation. One glucose molecule breaks down into two pyruvates (1). The energy from this exothermic reaction is used to bind inorganic phosphates to ADP and convert NAD+ to NADH. The two pyruvates are then broken down into two Acetaldehyde and give off two CO2 as a waste product (2). The two Acetaldehydes are then converted to two ethanol by using the H+ ions from NADH; converting NADH back into NAD+ (3).