Chapter: The Cell Membrane
Introduction to Lipids
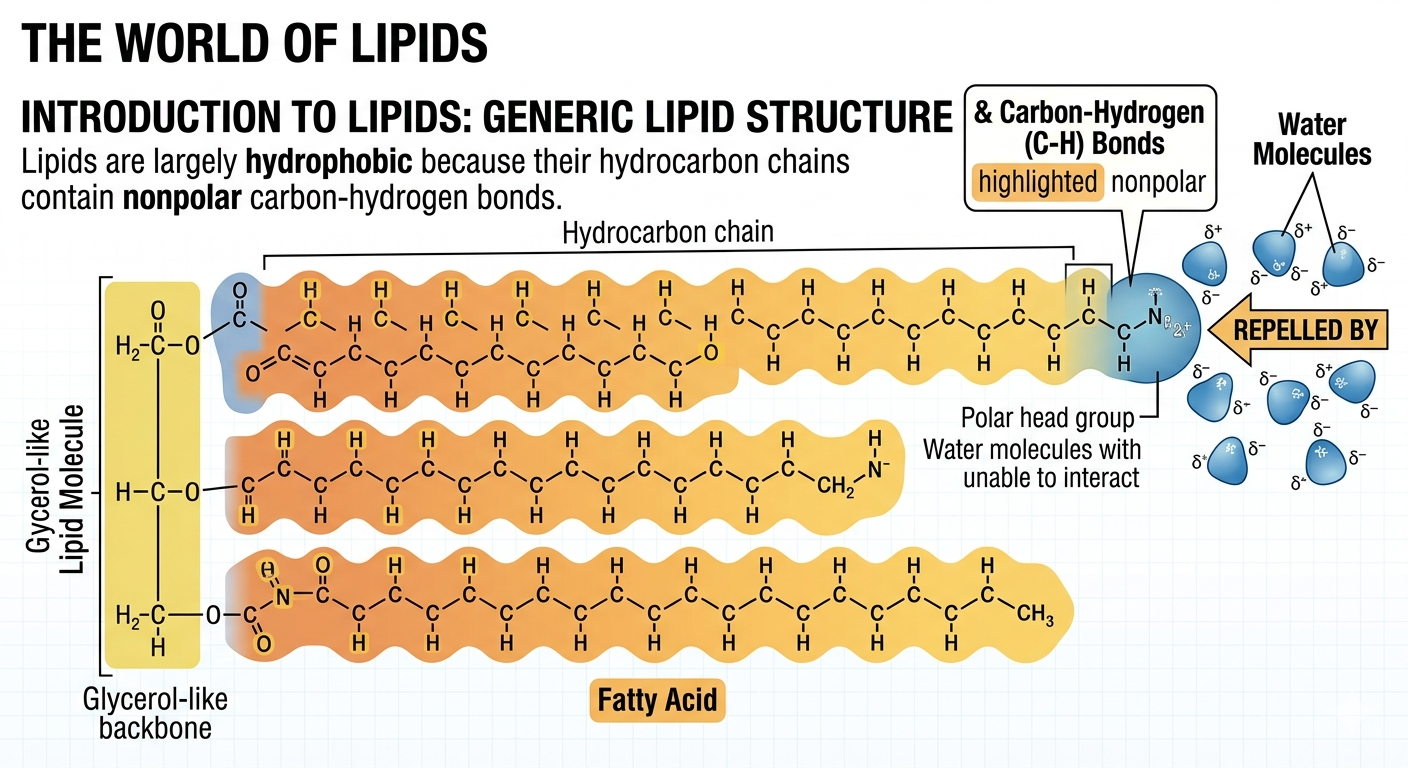
Lipids are a diverse group of biological molecules that share one important characteristic: at least part of the molecule has little or no affinity for water. In other words, lipids are hydrophobic. This hydrophobic nature results primarily from long chains of carbon and hydrogen atoms known as fatty acid chains. Carbon-hydrogen bonds are nonpolar, meaning electrons are shared relatively equally between the atoms, preventing the molecule from interacting favorably with polar water molecules. Although lipids vary greatly in structure and function, their hydrophobic properties are central to their biological roles. Lipids are essential for long-term energy storage, membrane structure, insulation, signaling, and cellular communication.
Figure 1. Lipids are largely hydrophobic because their hydrocarbon chains contain nonpolar carbon-hydrogen bonds.
Groups of Lipids
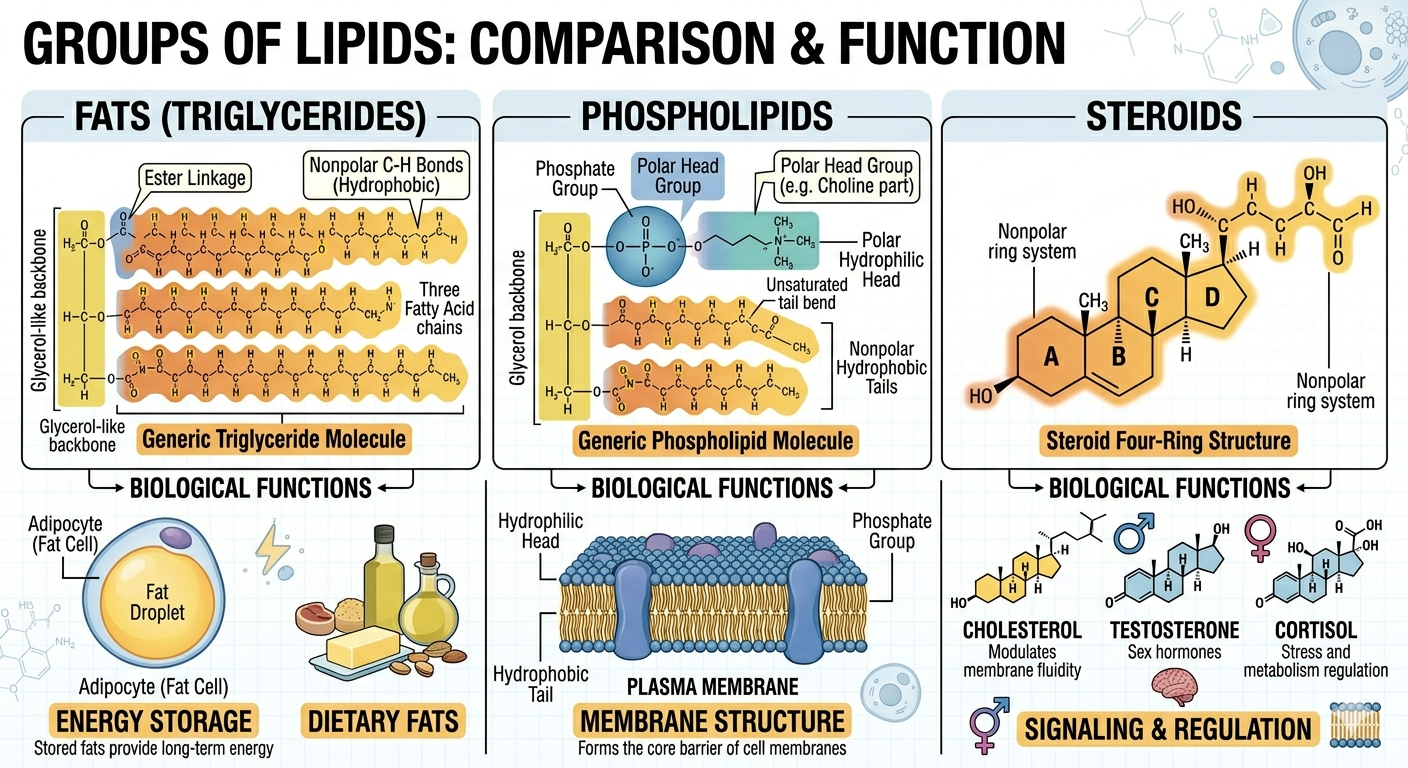
There are three major groups of lipids commonly discussed in biology: fats, phospholipids, and steroids. Although these molecules differ structurally, they all contain substantial nonpolar regions that make them hydrophobic. Fats primarily function in long-term energy storage, phospholipids form the structural basis of cell membranes, and steroids often function in signaling and membrane stability. Together, these lipid groups are essential for maintaining life.
Figure 2. The three major lipid groups differ in structure and function but all contain substantial hydrophobic regions.
Fats
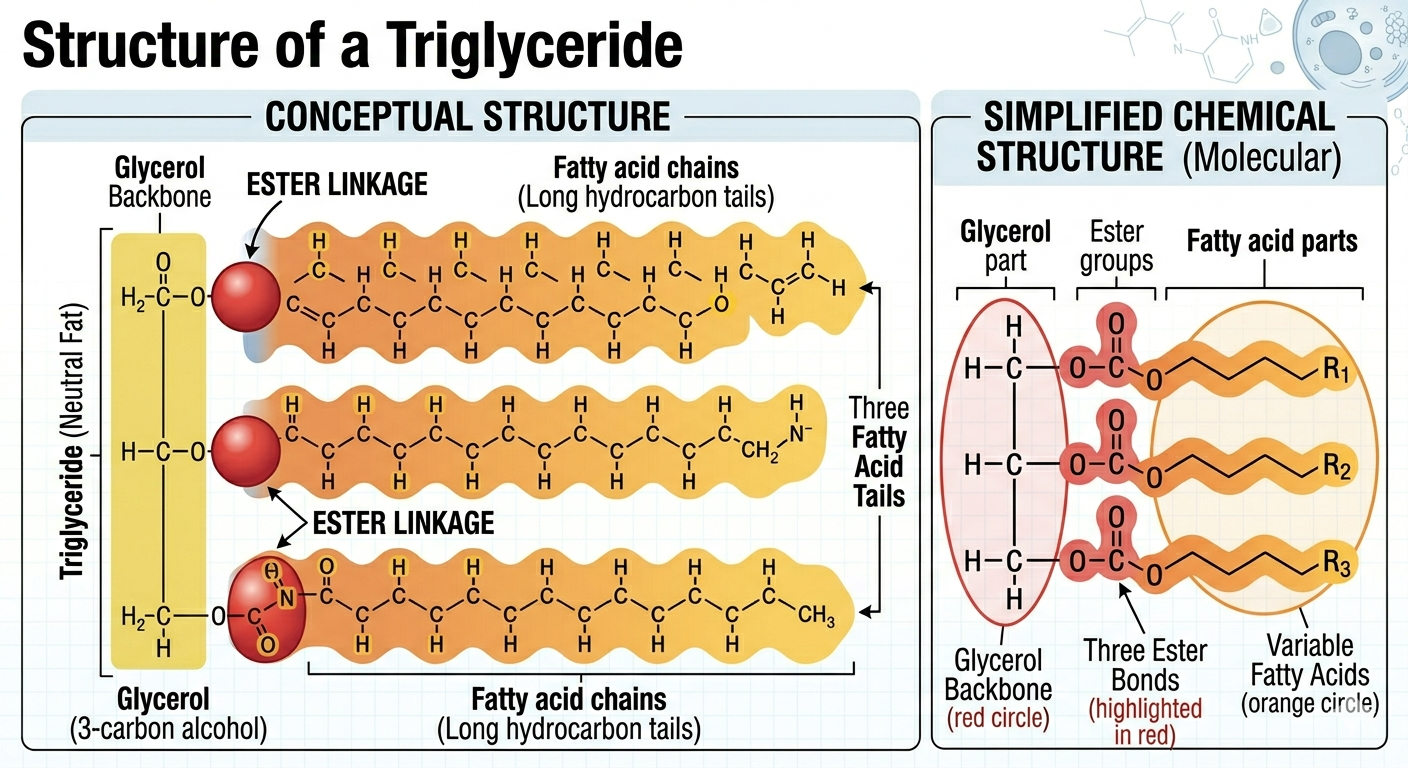
Fats are molecules specialized for long-term energy storage. A typical fat consists of a glycerol molecule attached to one or more fatty acid chains. In triglycerides, which are the most common fats in organisms, one glycerol molecule is attached to three fatty acid chains. The fatty acid chains contain numerous carbon-hydrogen bonds that store large amounts of chemical energy. When these bonds are broken during cellular respiration, the energy released can be used to produce ATP, the universal energy currency of the cell. Because fats store more energy per gram than carbohydrates, they serve as highly efficient energy reserves. In addition to energy storage, fats also function in insulation and cushioning of organs.
Figure 3. Triglycerides consist of glycerol attached to three fatty acid chains and function primarily in long-term energy storage.
Saturated and Unsaturated Fats
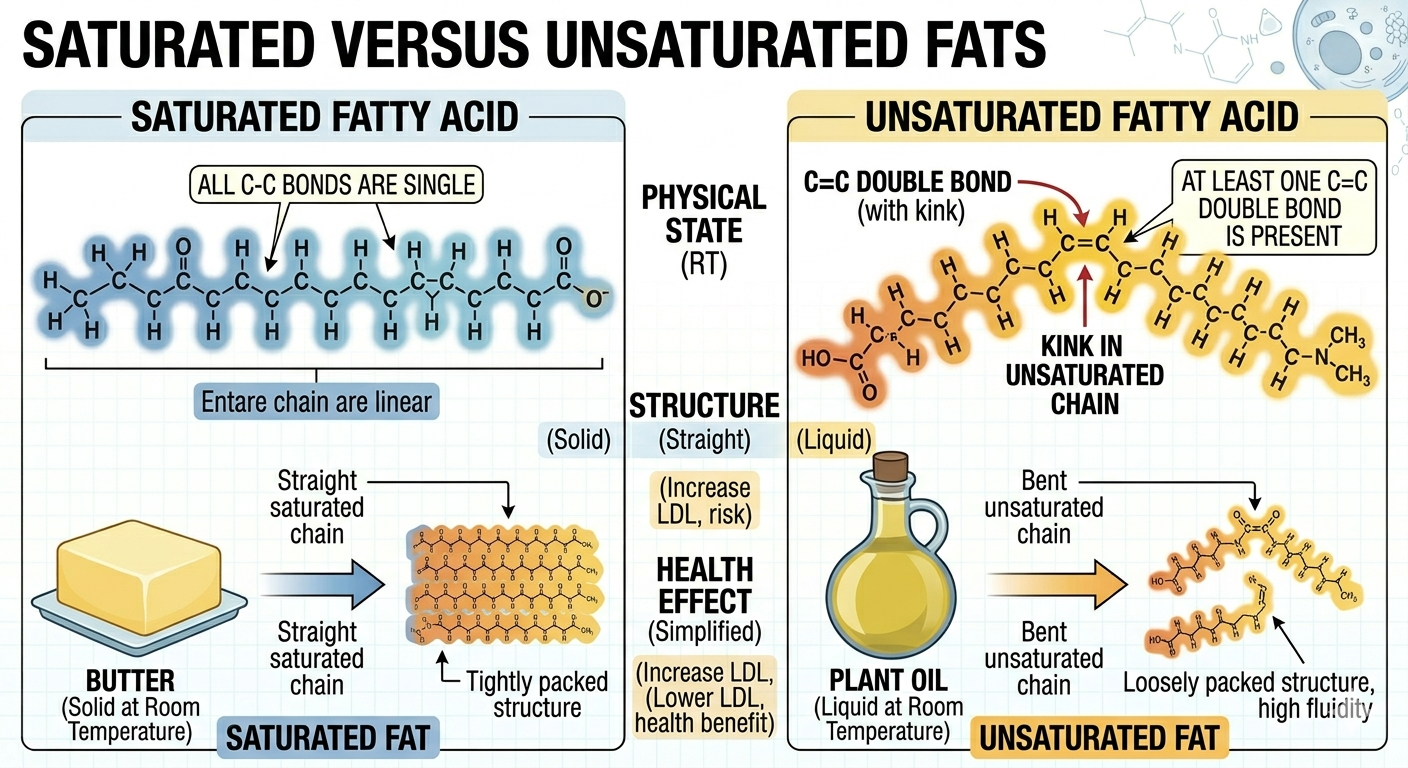
Fatty acids may be classified as saturated or unsaturated depending on the presence of carbon-carbon double bonds. Saturated fats contain fatty acid chains with only single bonds between carbon atoms. Because the chains remain straight, saturated fats can pack tightly together, making them solid at room temperature. Butter and animal fats are common examples. Unsaturated fats contain one or more double bonds within the fatty acid chain. These double bonds create bends, or kinks, in the molecule that prevent tight packing. As a result, unsaturated fats are usually liquid at room temperature and are commonly called oils. Monounsaturated fats contain one double bond, whereas polyunsaturated fats contain multiple double bonds. Unsaturated fats are generally considered healthier than saturated fats because they are metabolized more easily and are associated with lower risks of cardiovascular disease.
Figure 4. Double bonds in unsaturated fats create bends that prevent tight packing and increase fluidity.
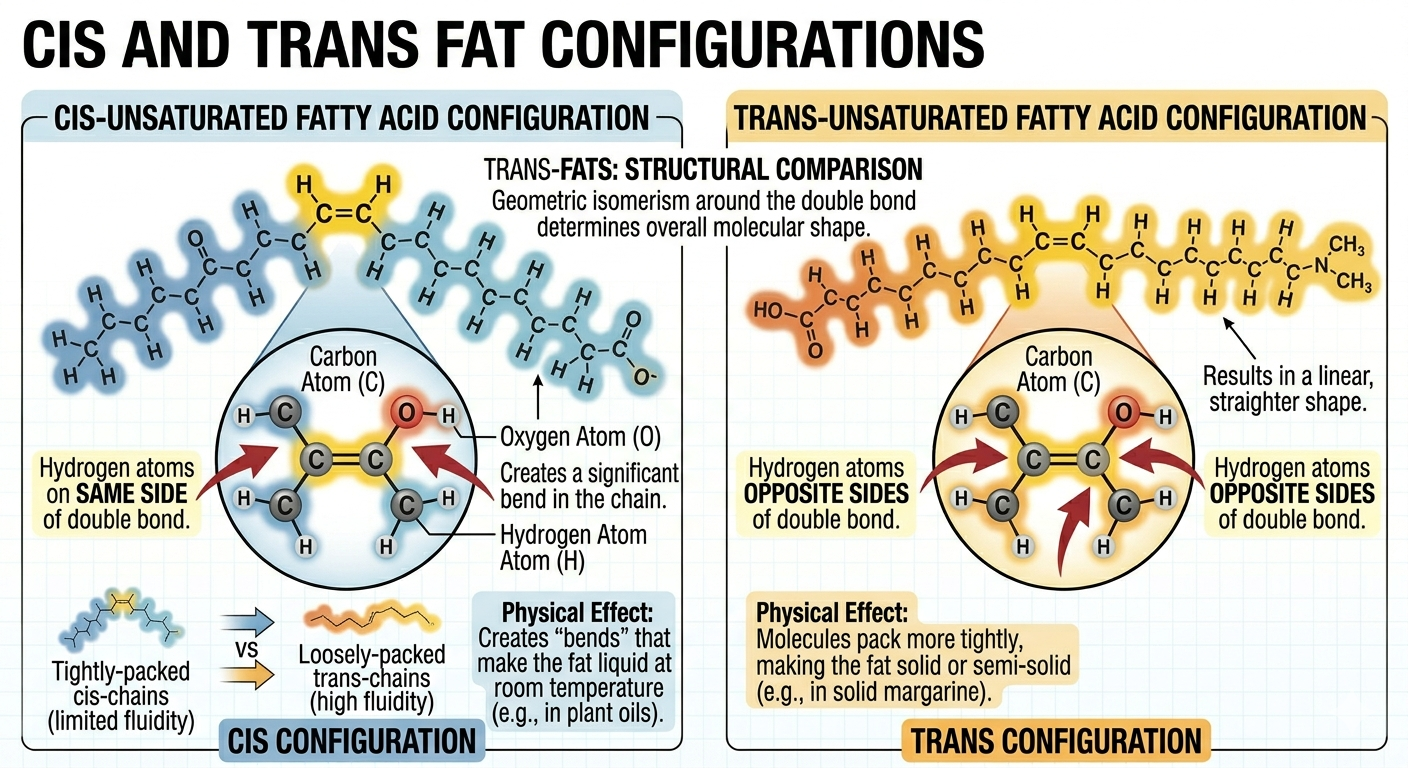
Cis and Trans Fats
Double bonds in unsaturated fats can exist in different orientations known as cis and trans configurations. In cis fats, the hydrogen atoms associated with the double bond are located on the same side of the molecule, creating a bend in the fatty acid chain. This bend prevents tight packing and helps maintain membrane fluidity. Most naturally occurring unsaturated fats are cis fats. In trans fats, however, the hydrogen atoms are positioned on opposite sides of the double bond, producing a straighter molecule that behaves more like a saturated fat. Trans fats are often created artificially during industrial hydrogenation of oils. Because trans fats pack more tightly and are more difficult for the body to metabolize, they are associated with increased risks of heart disease and other health problems.
Figure 5. Cis fats contain bends that increase fluidity, whereas trans fats are straighter and behave more like saturated fats.
Phospholipids
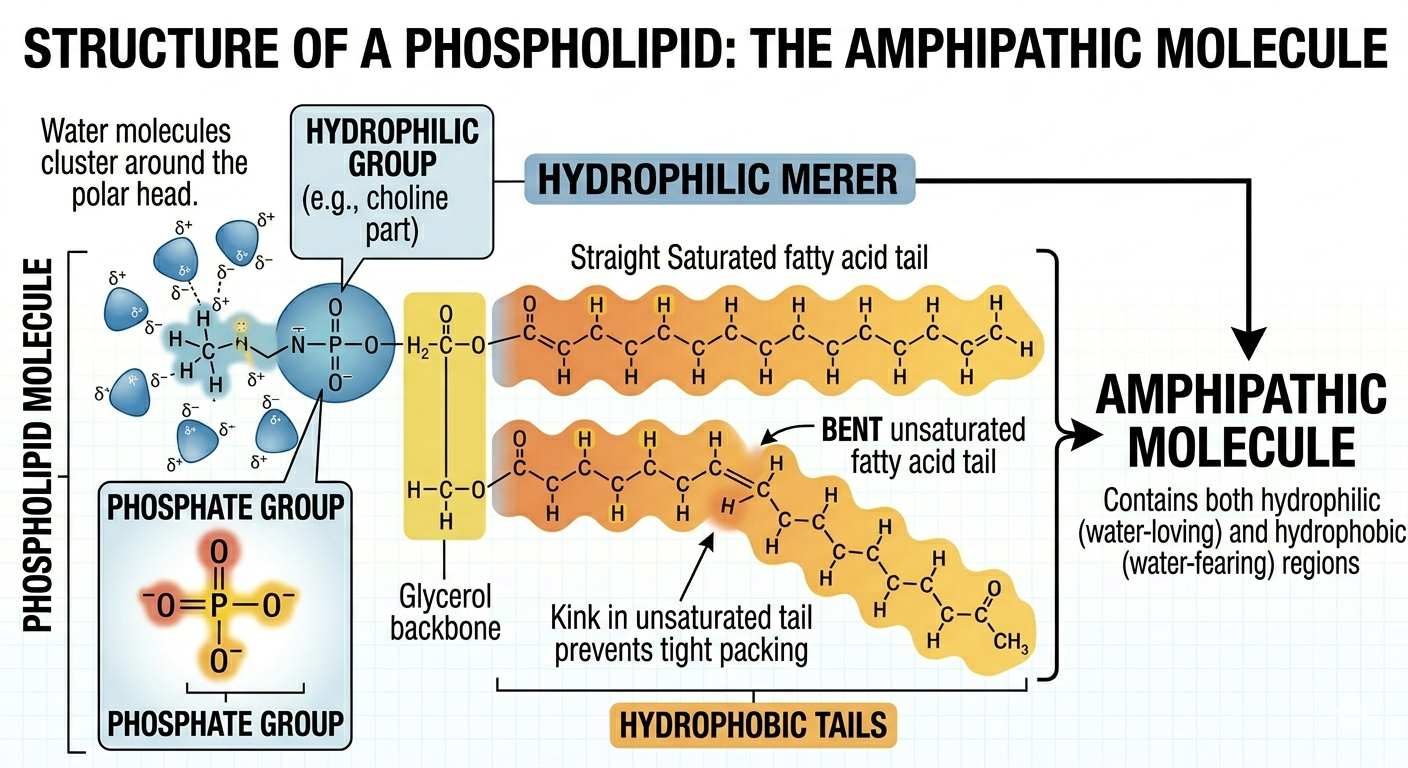
Phospholipids are a specialized group of lipids that form the structural foundation of cell membranes. A phospholipid consists of a glycerol molecule attached to two fatty acid tails and a phosphate-containing head group. The fatty acid tails are hydrophobic and avoid water, whereas the phosphate head is polar and hydrophilic, meaning it interacts favorably with water. Because phospholipids contain both hydrophobic and hydrophilic regions, they are described as amphipathic molecules. One of the fatty acid tails commonly contains a double bond that creates a bend, helping maintain membrane flexibility and fluidity.
Figure 6. Phospholipids possess both hydrophilic and hydrophobic regions making them amphipathic molecules.
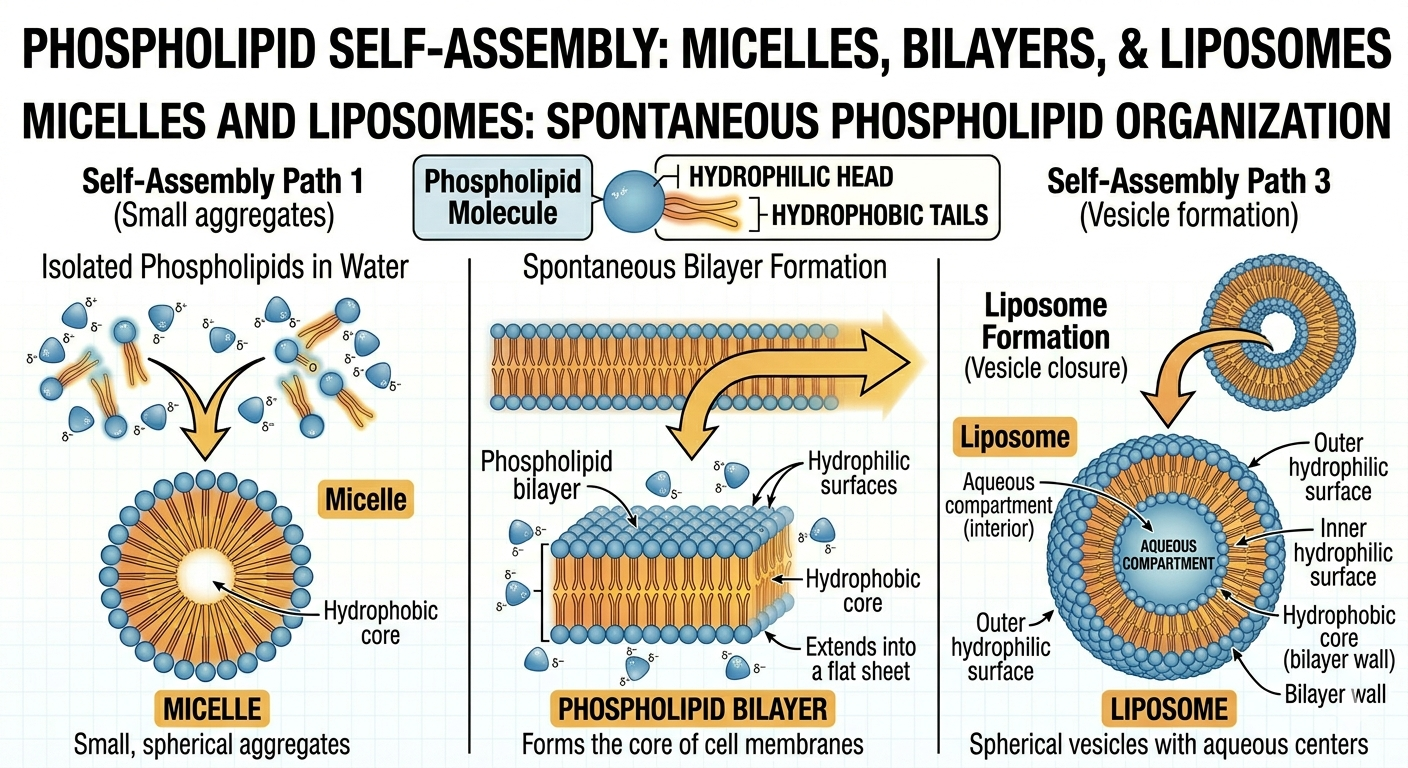
Formation of Lipid Bilayers
When phospholipids are placed in water, they spontaneously organize into structures that minimize contact between hydrophobic tails and water molecules. One possible structure is a micelle, in which the hydrophilic heads face outward toward the water while the hydrophobic tails point inward. Another more biologically important structure is the phospholipid bilayer. In a bilayer, two layers of phospholipids arrange themselves tail-to-tail with hydrophilic heads facing the surrounding water on both sides. This arrangement forms the basis of all cell membranes. In some cases, phospholipid bilayers form spherical structures known as liposomes or vesicles, which can enclose water and dissolved substances.
Figure 7. Phospholipids spontaneously organize into structures that minimize contact between hydrophobic tails and water.
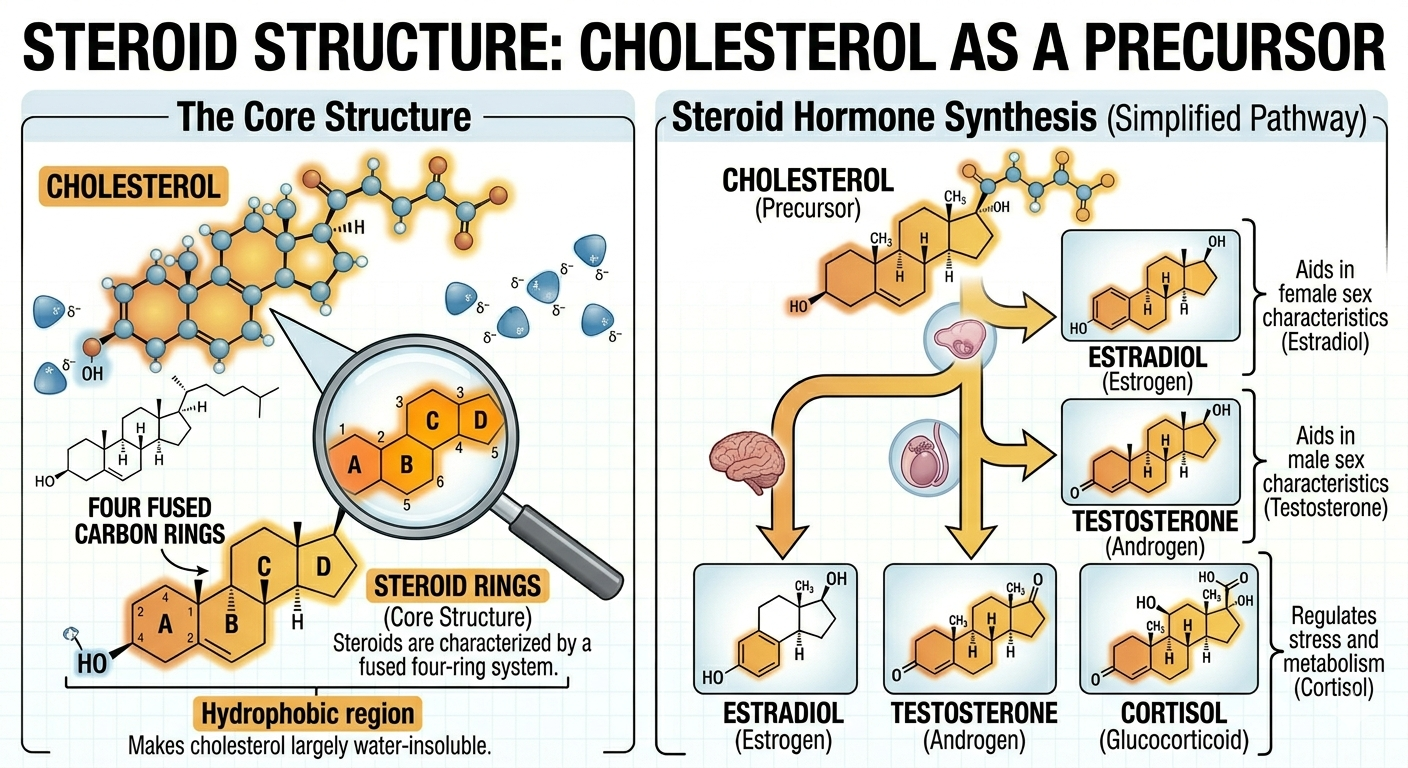
Steroids
Steroids are lipids characterized by a carbon skeleton consisting of four fused carbon rings. Although steroids differ from fats and phospholipids in structure, they remain largely hydrophobic due to their extensive nonpolar regions. Cholesterol is one of the most important steroids in animals and serves as the precursor for many other steroid molecules including testosterone, estrogen, cortisol, and other hormones. Steroid hormones function as chemical messengers that regulate growth, metabolism, reproduction, and numerous physiological processes. Cholesterol also plays an important structural role within cell membranes by helping regulate membrane fluidity.
Figure 8. Steroids contain four fused carbon rings and often function in membrane stability and cellular signaling.
The Cell Membrane
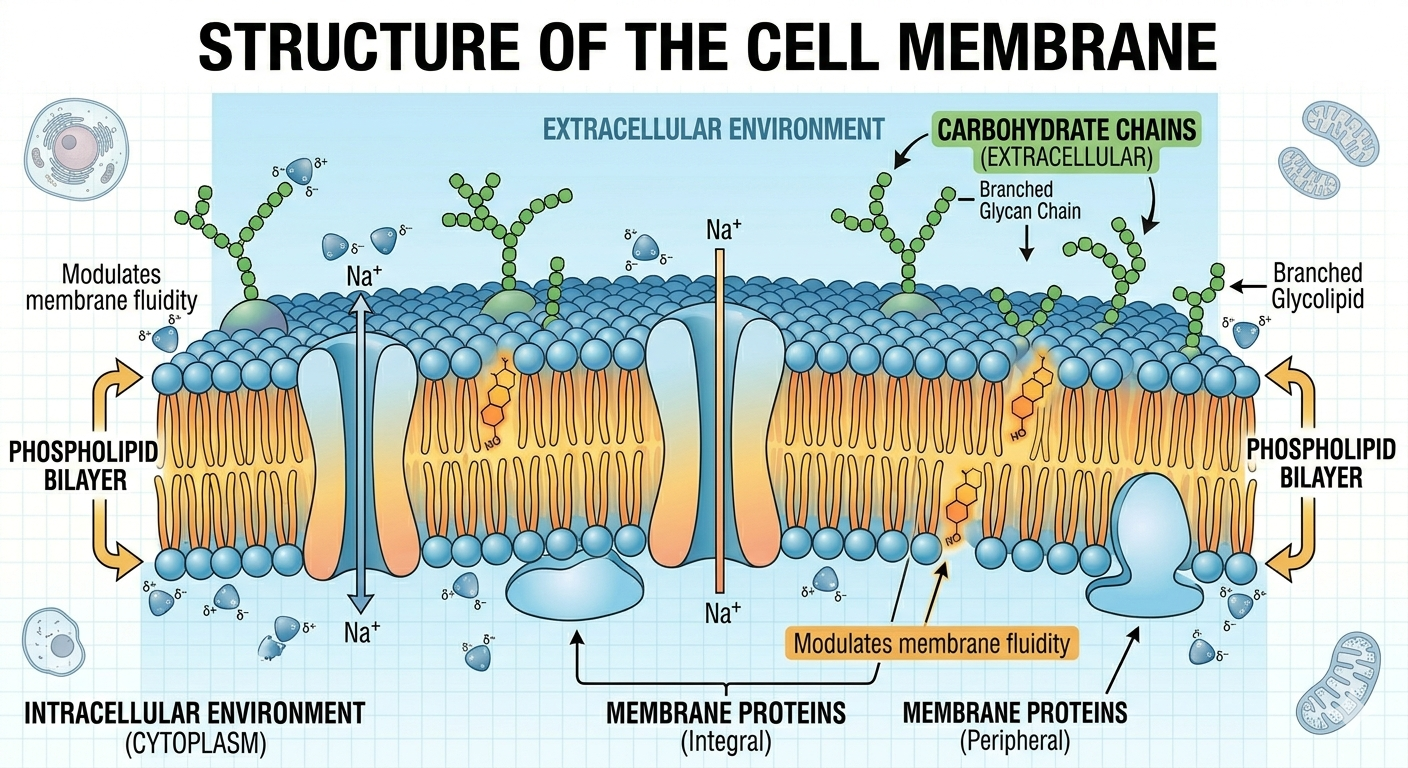
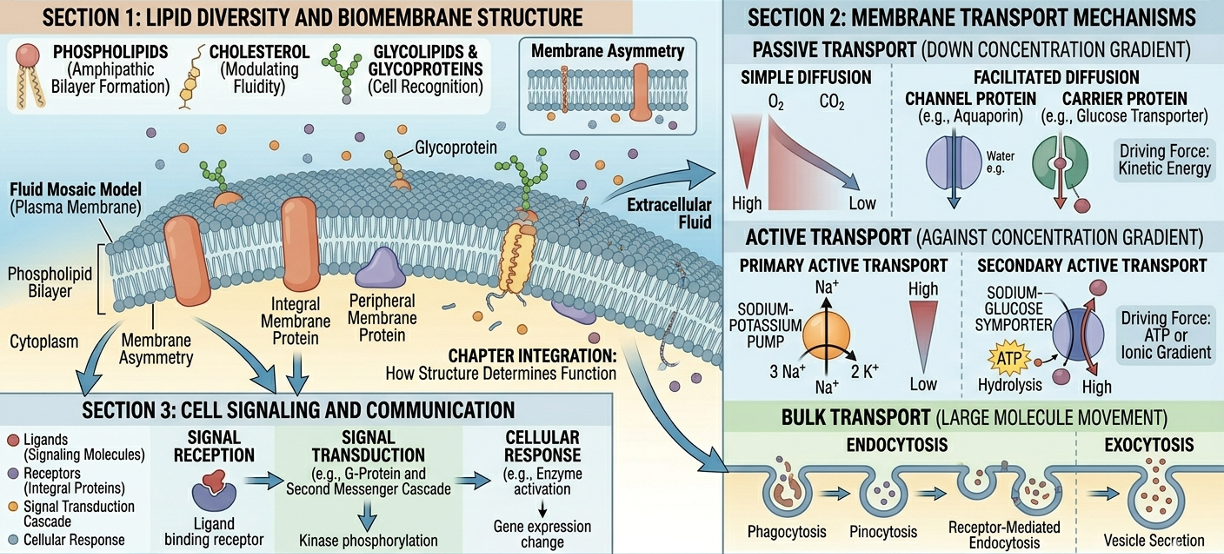
The cell membrane, also known as the plasma membrane, is the boundary that separates the internal environment from the outside world. The membrane consists primarily of a phospholipid bilayer with embedded proteins, cholesterol, and carbohydrates. The membrane is selectively permeable, meaning it allows some substances to cross easily while restricting others. Small nonpolar molecules such as oxygen and carbon dioxide diffuse readily through the membrane, whereas larger or charged molecules often require specialized transport proteins. The cell membrane is essential for maintaining homeostasis by regulating the movement of materials into and out of the cell.
Figure 9. The cell membrane consists primarily of a phospholipid bilayer with embedded proteins and other molecules.
The Fluid Mosaic Model
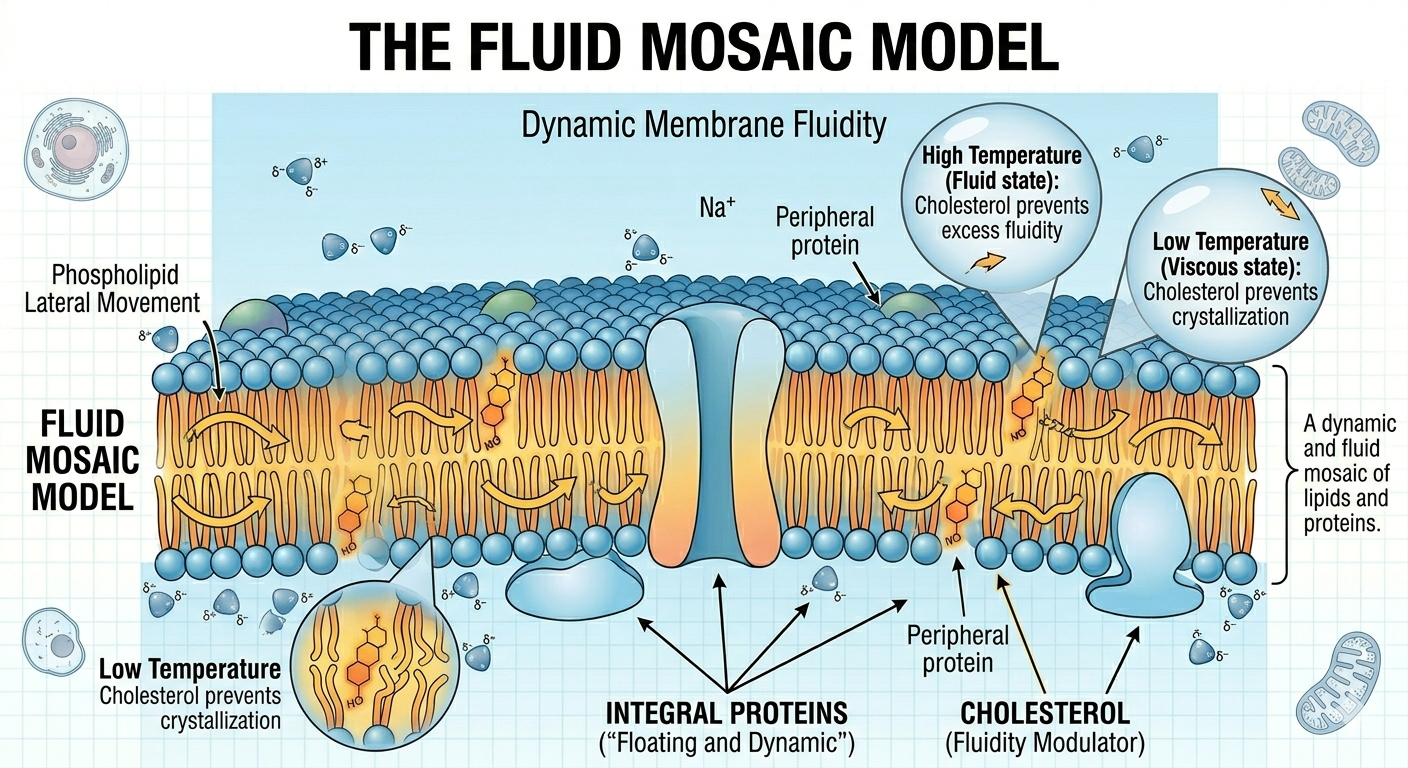
The modern model of membrane structure is known as the fluid mosaic model. According to this model, the phospholipid bilayer behaves like a fluid in which proteins and other molecules can move laterally. Embedded proteins float within the membrane much like boats floating on water. Some proteins extend completely through the membrane and function in transport, signaling, or enzymatic activity, whereas others are attached only to one side of the membrane. Cholesterol molecules are interspersed among the phospholipids and help stabilize membrane structure. The term “mosaic” refers to the wide variety of molecules embedded within the membrane.
Figure 10. The fluid mosaic model describes membranes as dynamic structures in which molecules move laterally within the bilayer.
Membrane Proteins
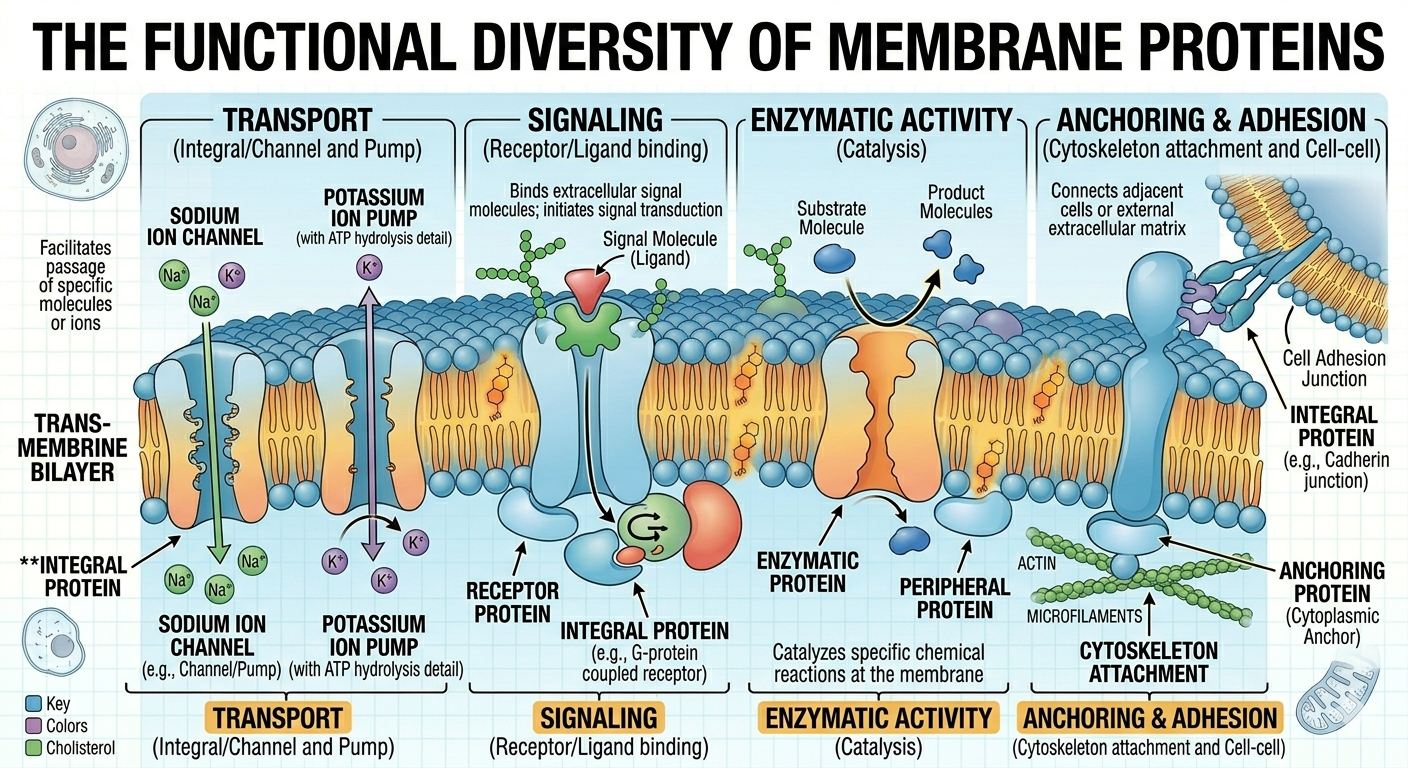
Membrane proteins perform a wide variety of essential cellular functions and are critical for maintaining cellular homeostasis. Transport proteins regulate the movement of substances across the membrane, allowing nutrients, ions, and waste products to enter or leave the cell. Receptor proteins bind signaling molecules such as hormones or neurotransmitters and trigger intracellular responses that alter cellular activity. Enzymatic proteins embedded within the membrane catalyze chemical reactions at the membrane surface, often participating in metabolic pathways or signal transduction. Some membrane proteins anchor the cytoskeleton to the membrane, helping maintain cell shape and structural stability, while others participate in cell recognition and communication between neighboring cells. Integral proteins extend partially or completely through the phospholipid bilayer, whereas peripheral proteins are attached loosely to the inner or outer membrane surface. Many membrane proteins are highly specific and interact only with certain molecules, functioning much like a lock that recognizes only a specific key. Glycoproteins, which are proteins with attached carbohydrate chains, serve as cellular identification markers and play critical roles in immune system recognition and tissue organization. Collectively, membrane proteins allow cells to communicate, transport materials, respond to environmental changes, and coordinate activities throughout multicellular organisms.
Figure 11. Membrane proteins perform transport, signaling, enzymatic, and structural functions.
Membrane Permeability
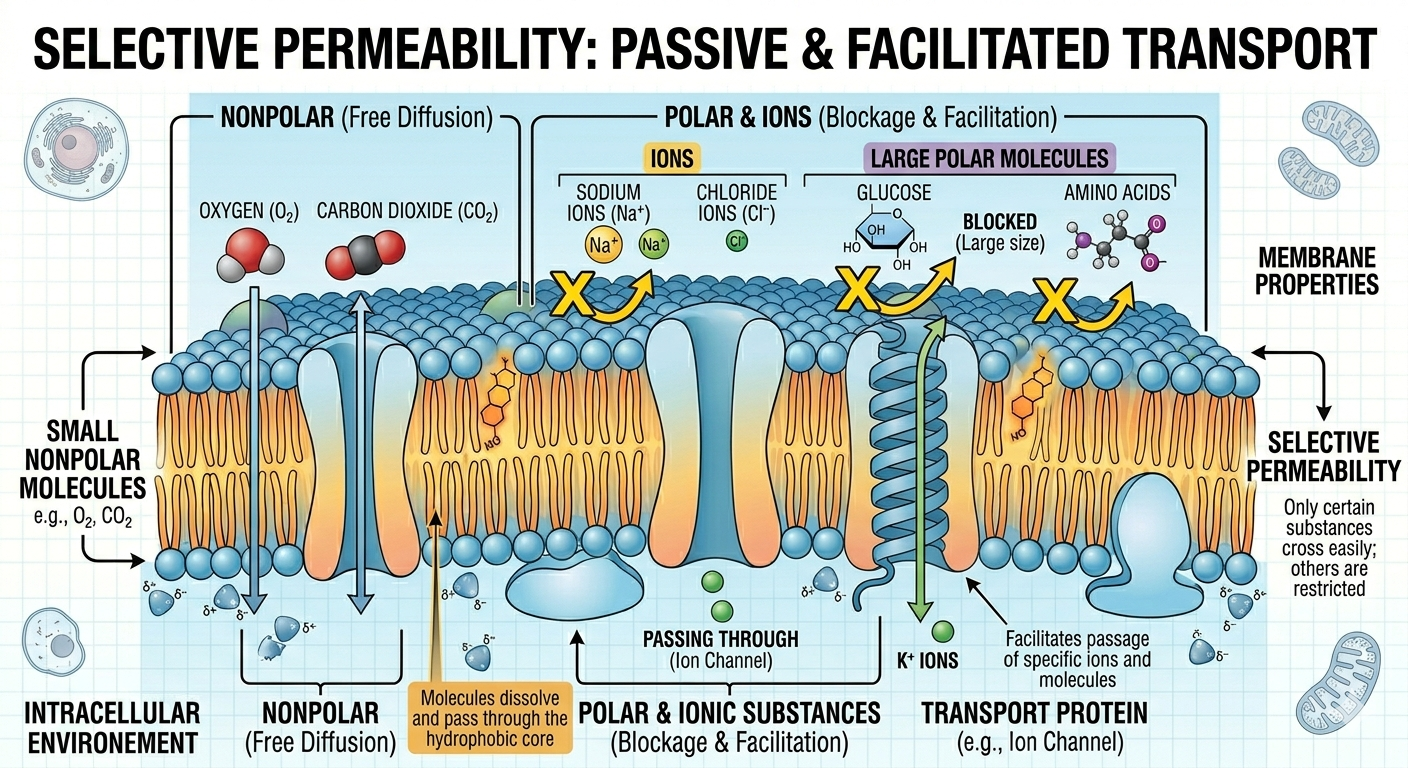
Membrane permeability refers to how easily substances can cross the cell membrane. The phospholipid bilayer allows small nonpolar molecules to diffuse relatively freely because they can move through the hydrophobic interior of the membrane. In contrast, ions and large polar molecules have difficulty crossing the membrane because they are repelled by the nonpolar fatty acid tails. As a result, many substances require transport proteins to enter or leave the cell. Membrane permeability is critical for maintaining proper concentrations of ions, nutrients, and water inside the cell.
Figure 12. Cell membranes allow some substances to cross easily while restricting others.
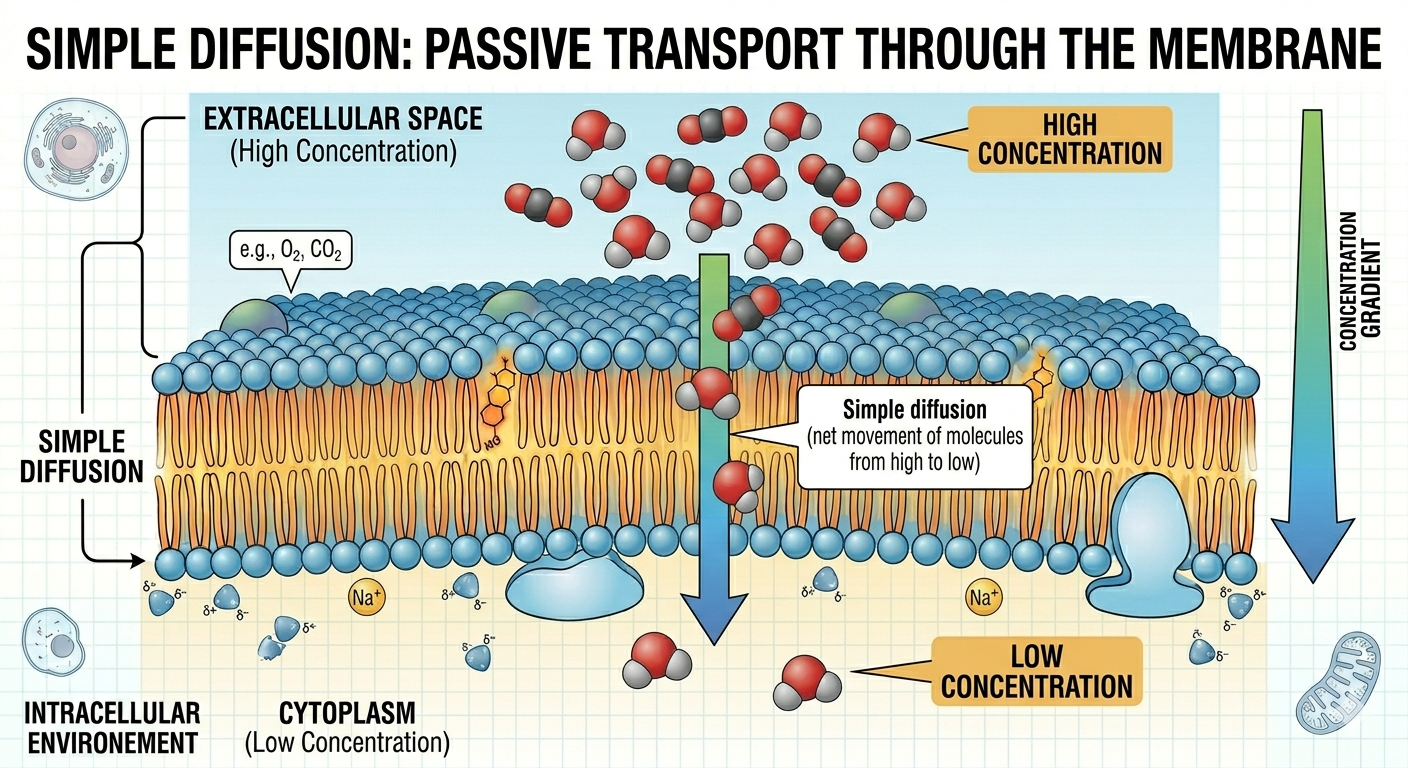
Passive Transport
Passive transport is the movement of substances across the cell membrane without the expenditure of cellular energy. In passive transport, substances move down their concentration gradient from regions of higher concentration to regions of lower concentration. One form of passive transport is simple diffusion, in which molecules move directly through the membrane. Very small molecules, such as oxygen and carbon dioxide, commonly move across the membrane through simple diffusion.
Figure 13. Simple diffusion allows small nonpolar molecules to move directly through the membrane down their concentration gradient.
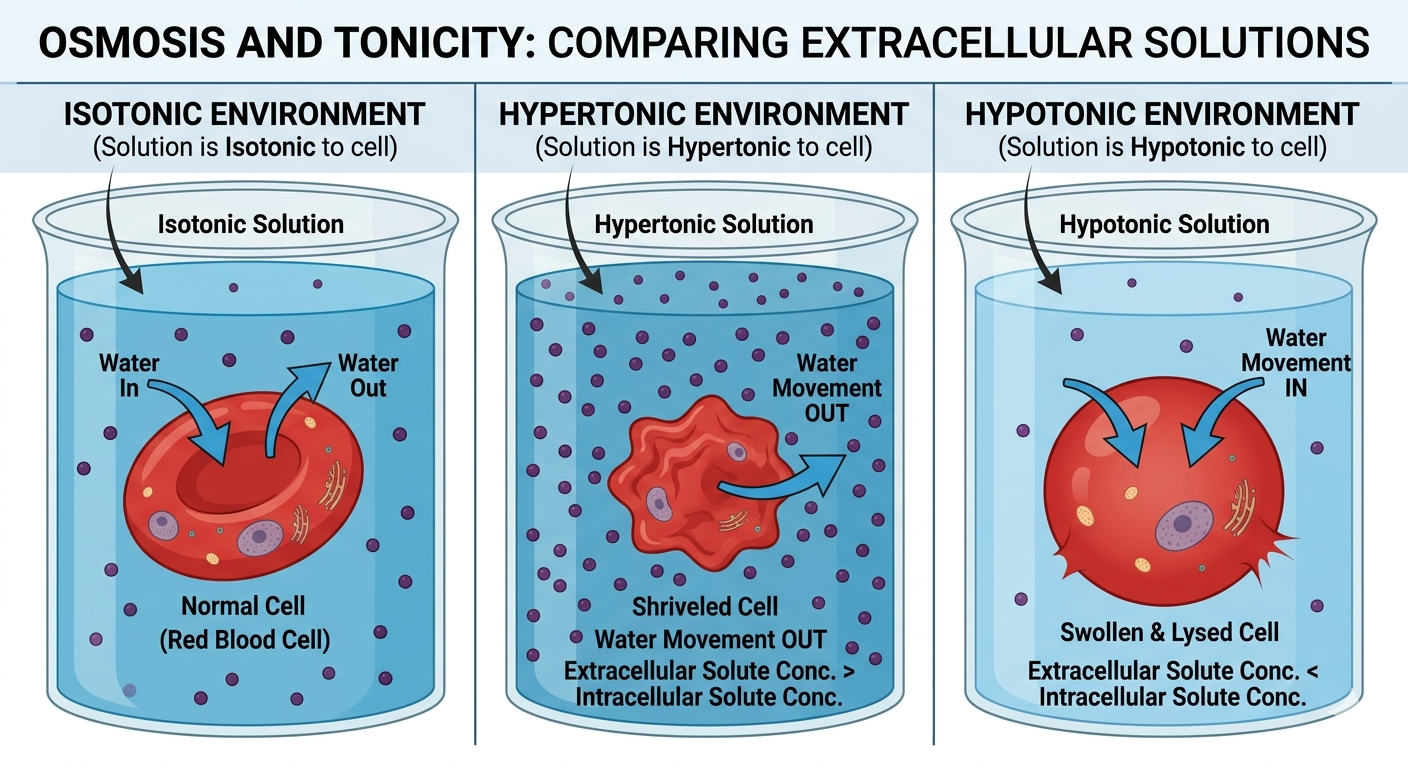
Osmosis
Osmosis is the diffusion of water across a selectively permeable membrane. Water moves from regions with lower solute concentration (high water concentration) to regions with higher solute concentration (low water concentration). Osmosis plays a major role in maintaining cell volume and fluid balance. In an isotonic environment, solute concentrations are equal inside and outside the cell, so water movement is balanced. In a hypertonic environment, the surrounding solution contains a higher solute concentration than the cell, causing water to leave the cell and resulting in cell shrinkage. In a hypotonic environment, the surrounding solution contains a lower solute concentration than the cell, causing water to enter the cell and potentially leading to swelling or bursting. Proper osmotic balance is essential for normal cellular function.
Figure 14. Osmosis regulates water movement across membranes and strongly influences cell volume.
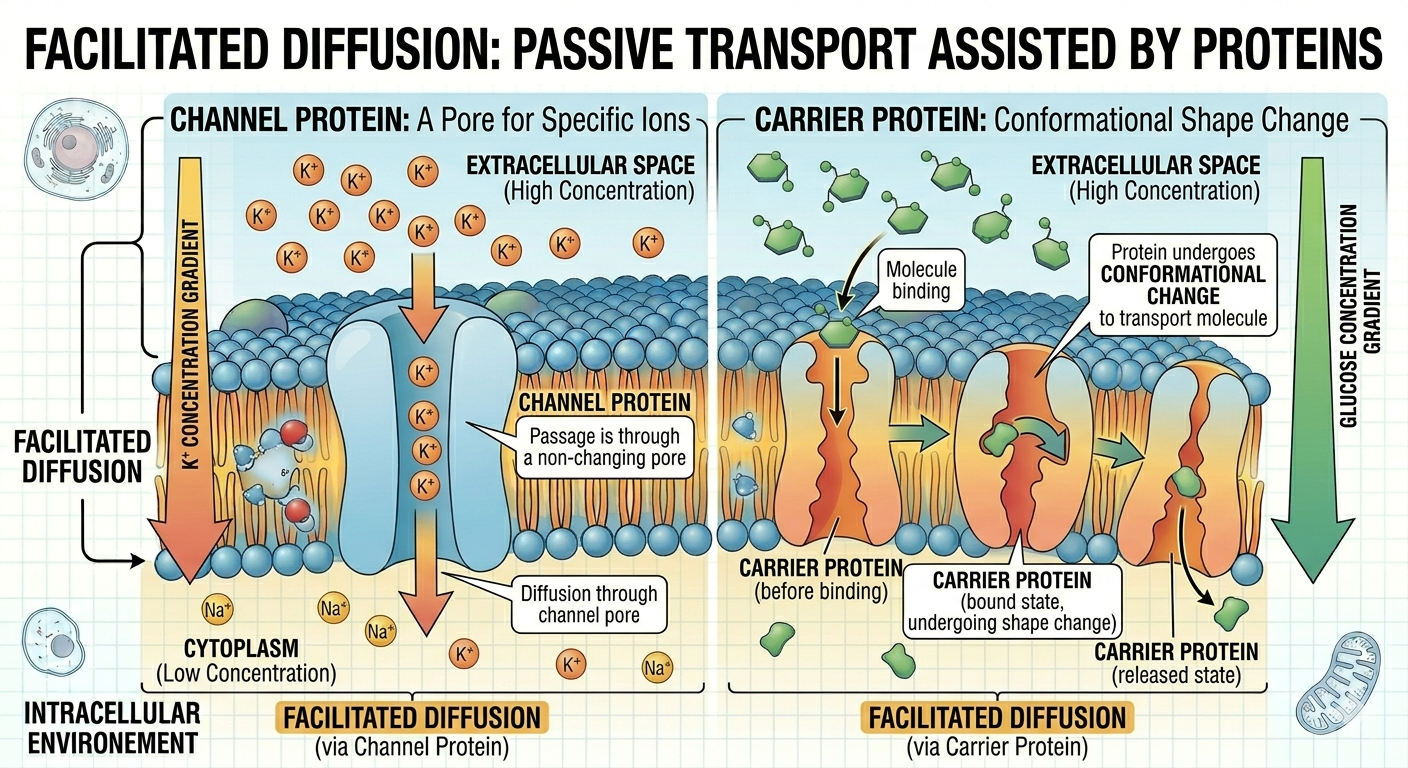
Facilitated diffusion
Another important form of passive transport is facilitated diffusion, a process in which membrane transport proteins help substances cross the cell membrane when they cannot pass directly through the phospholipid bilayer on their own. Unlike simple diffusion, facilitated diffusion is necessary for many ions, polar molecules, and larger substances such as glucose because these materials are unable to move through the hydrophobic interior of the membrane independently. Channel proteins create selective openings that allow specific ions or molecules to move across the membrane, while carrier proteins bind to particular substances and undergo shape changes that transport them from an area of higher concentration to an area of lower concentration. Because facilitated diffusion moves substances down their concentration gradient, the process does not require cellular energy in the form of ATP. This mechanism is essential for maintaining cellular homeostasis and regulating the movement of nutrients, ions, and other important molecules into and out of cells.
Figure 14. Facilitated diffusion uses transport proteins to move substances down their concentration gradients without ATP.
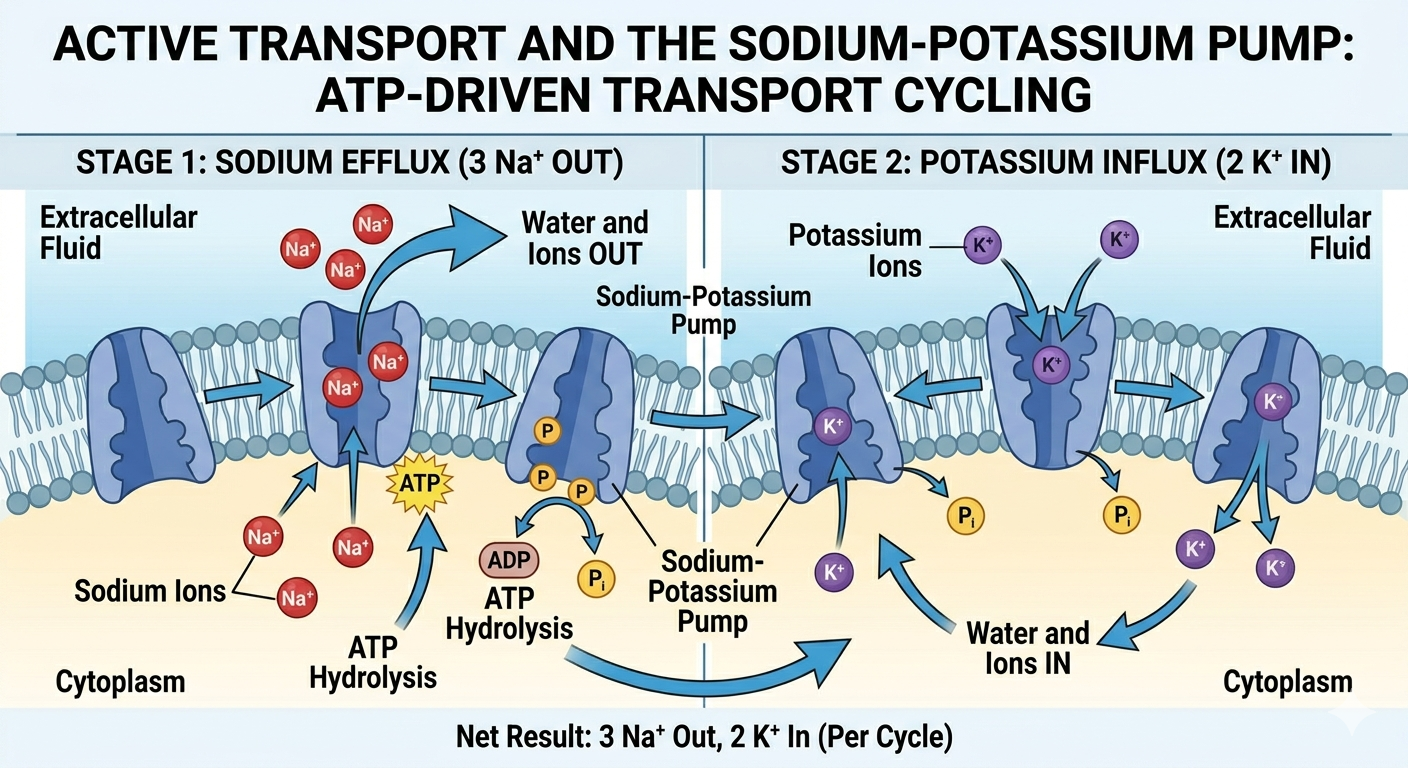
Active Transport
Active transport requires cellular energy, usually in the form of ATP, to move substances across the membrane against their concentration gradient. In active transport, substances move from regions of lower concentration to regions of higher concentration. One of the best-known examples is the sodium-potassium pump. This membrane protein uses ATP to pump sodium ions out of the cell and potassium ions into the cell. Specifically, the pump transports three sodium ions out of the cell and two potassium ions into the cell during each cycle. This unequal exchange helps maintain membrane potential, regulate osmotic balance, and support nerve and muscle function.
Figure 15. The sodium-potassium pump uses ATP to transport ions against their concentration gradients.
Bulk Transport
Some materials are too large to cross the membrane through transport proteins and instead move by bulk transport mechanisms involving vesicles.
Exocyotsis
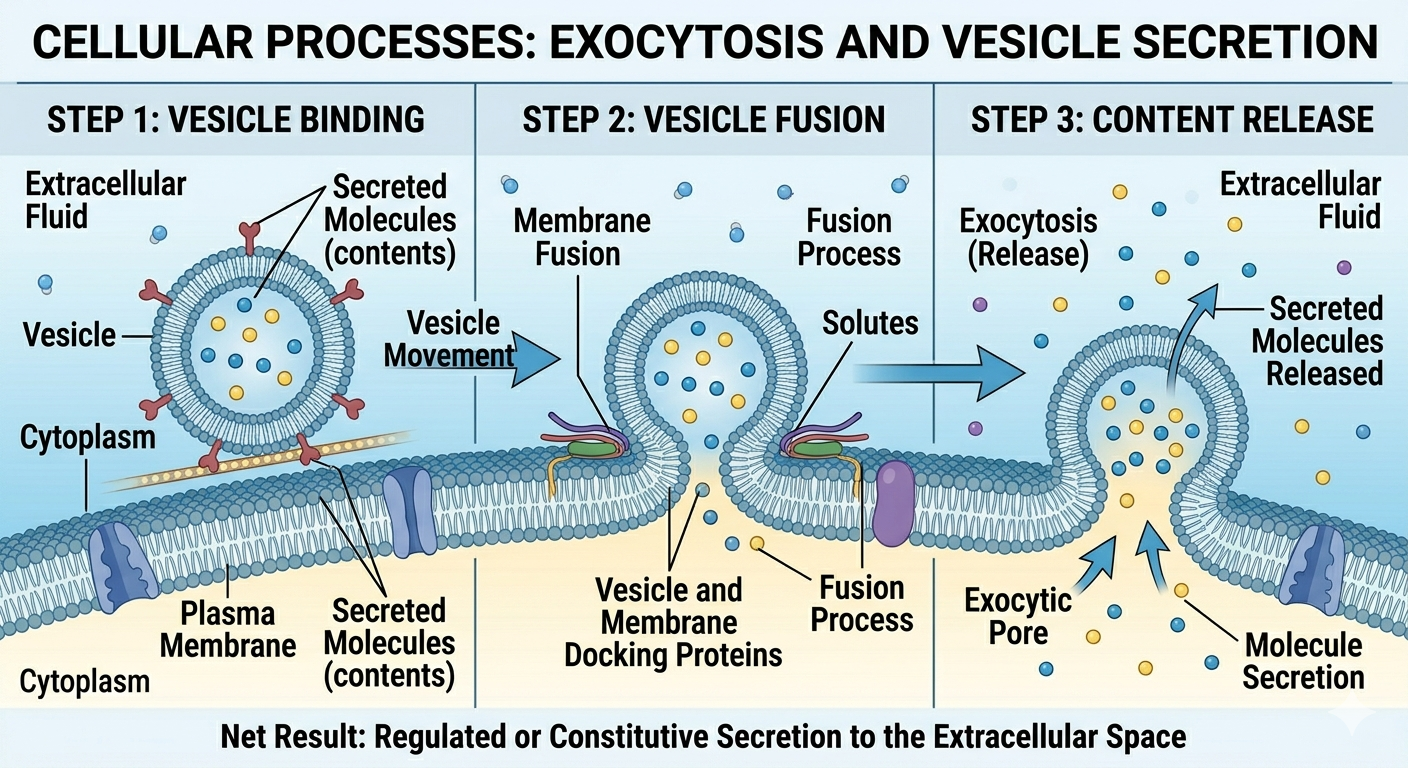
In exocytosis, intracellular secretory vesicles move toward the plasma membrane and fuse with it, allowing their contents to be released into the extracellular environment. This process is essential for cellular communication and export, enabling cells to secrete important molecules such as hormones, neurotransmitters, and enzymes, as well as eliminate waste materials that the cell no longer needs. Exocytosis is also tightly regulated, ensuring that these substances are released only when and where they are required for proper physiological function.
Figure 16. Exocytosis releases materials from the cell through vesicle fusion with the plasma membrane.
Endocyotsis
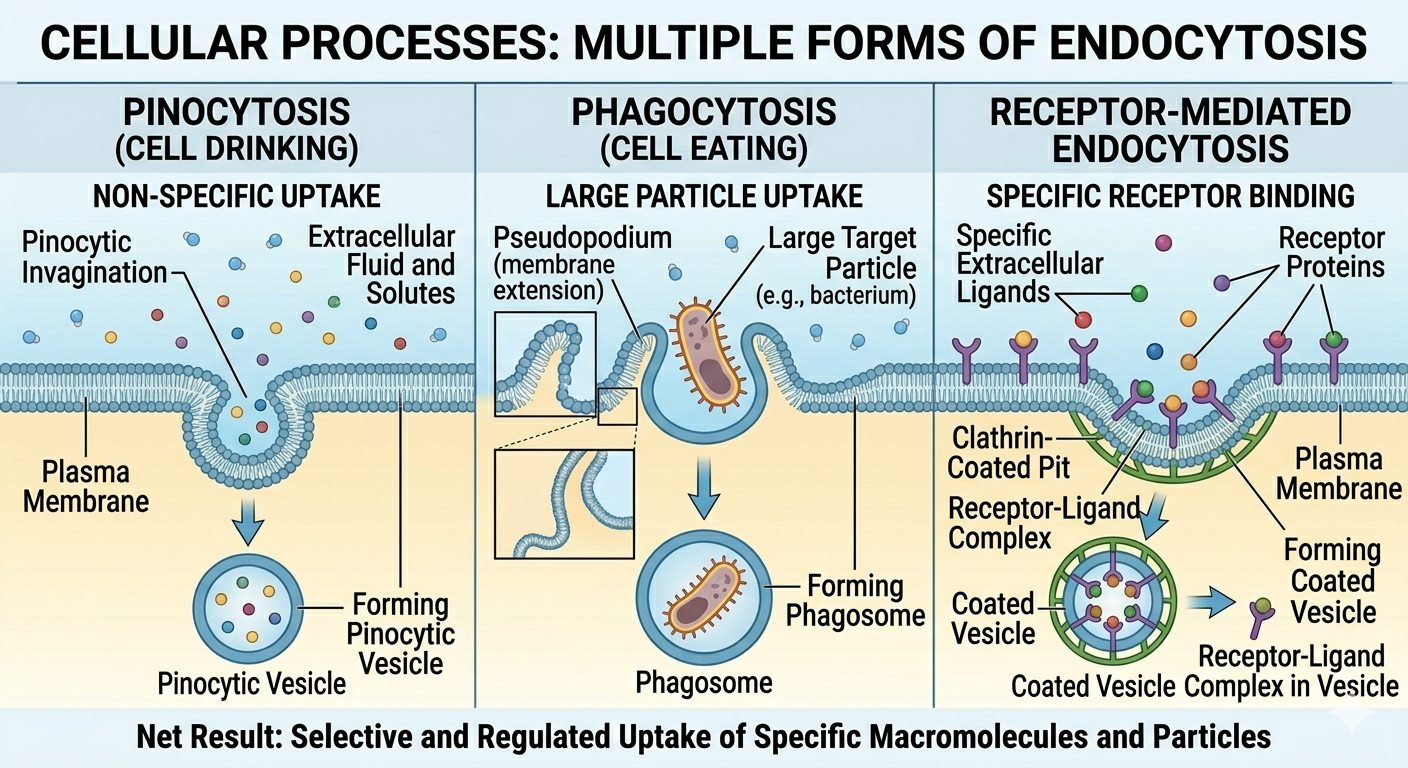
In endocytosis, the plasma membrane folds inward to surround external material, forming a vesicle that brings substances into the cell. One form, pinocytosis, involves the nonspecific uptake of extracellular fluid along with dissolved solutes, essentially allowing the cell to sample its surroundings. In contrast, receptor-mediated endocytosis is highly selective, importing only specific molecules that bind to specialized membrane receptors, ensuring that only targeted substances are taken in. These mechanisms enable cells to transport large or otherwise impermeable materials across the membrane while also playing a key role in regulating nutrient uptake, signaling, and environmental communication.
Figure 17. Endocytosis allows cells to internalize fluids, particles, and specific molecules using membrane vesicles.
The Origins of the First Cells
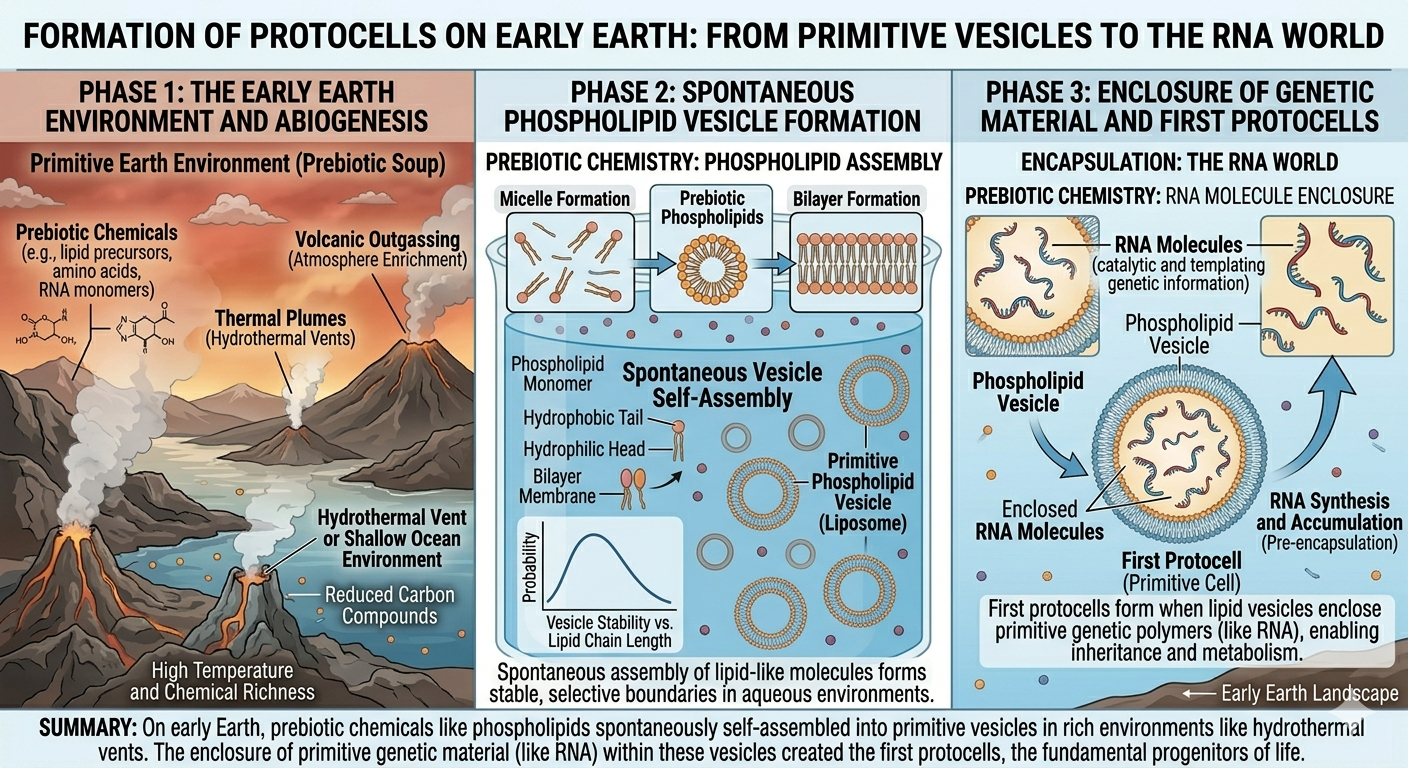
Scientists believe the first cells appeared approximately 4.0 to 4.3 billion years ago shortly after Earth formed. Several hypotheses attempt to explain the origin of the first cells, including deep-sea hydrothermal vent models, lightning-driven chemical synthesis, and the delivery of organic molecules by meteorites. Many researchers propose that RNA was the first self-replicating molecule in a stage referred to as the RNA world. The earliest cells were likely simple heterotrophs that relied on environmental sources of organic molecules for energy. One of the most important steps in the origin of life was the formation of membranes. Because phospholipids spontaneously form bilayers and vesicles in water, early membrane-like structures may have formed naturally on the prebiotic Earth. These primitive membranes could have enclosed self-replicating molecules such as RNA, creating protocells that eventually evolved into the first true cells.
Figure 18. Primitive membrane vesicles may have enclosed self-replicating molecules during the origin of the first cells.
The Importance of Lipids and Cell Membranes
Lipids and cell membranes are essential to all known forms of life, forming the structural and functional basis of cellular organization. Lipids serve multiple roles, including long-term energy storage, thermal insulation, cellular signaling, and the formation of biological membranes. Cell membranes, in turn, regulate the movement of substances into and out of the cell, maintaining homeostasis by controlling internal conditions despite external changes. The phospholipid bilayer provides both structural integrity and flexibility, enabling membranes to be stable yet dynamic enough to allow communication and transport. A strong understanding of lipid chemistry and membrane structure is therefore central to biology, medicine, physiology, and biochemistry, especially because disruptions in membrane function are linked to serious conditions such as cardiovascular disease, neurodegenerative disorders, and metabolic syndromes.
Figure 19. Lipids and cell membranes regulate structure, transport, communication, and homeostasis in living cells.